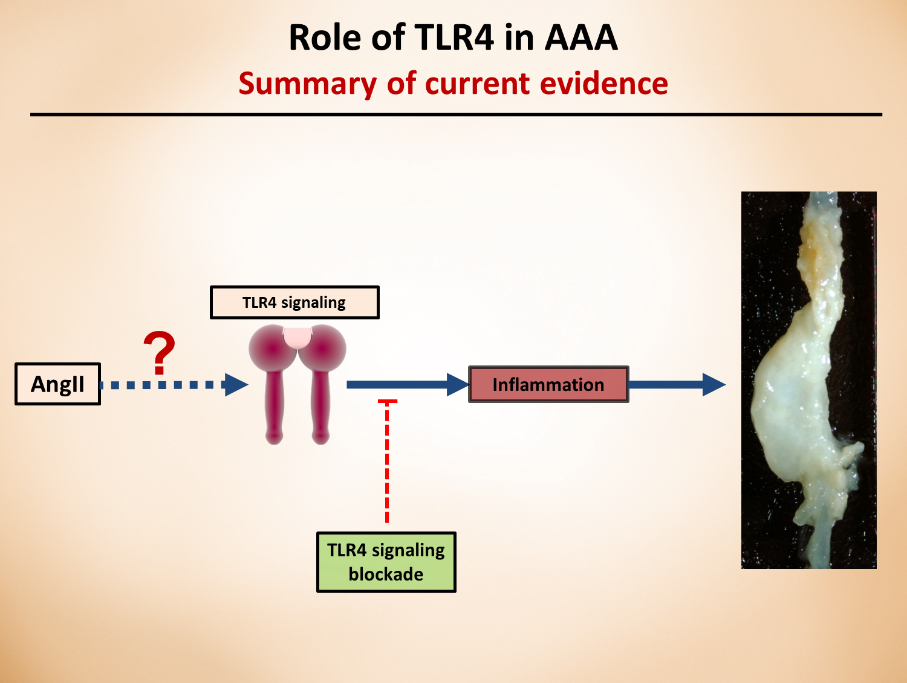
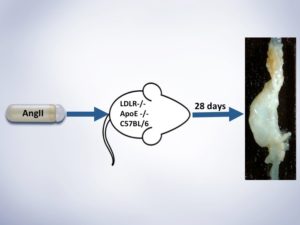
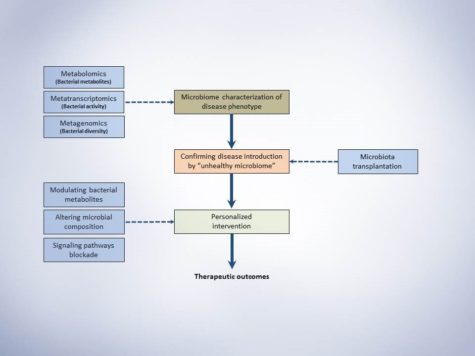
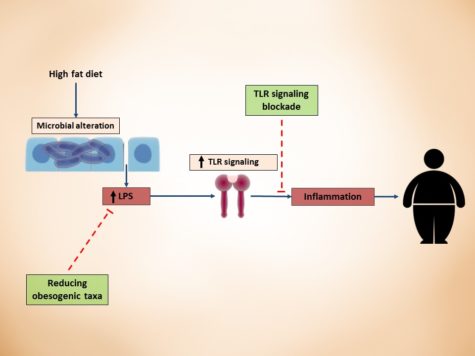
Fifth Year at Vascular Discovery: How an Early Career Navigates Through a Virtual Event
It is not news anymore that after World Health Organization (WHO) classified novel coronavirus disease (COVID-19) as a pandemic, the real-life impact of the “new normal” started to show itself. We started to see the impact by pausing the research and of course, cancelation of all scientific events. As scientists shifted their focus toward the data analysis, manuscript preparation, grants or any other means of remote work, the American Heart Association also shifted its focus to keep events happening, virtually. This new mode of attending a conference has many perks including petting your dog when the field’s connoisseur is giving a talk, drinking coffee from your favorite mug, or keeping the PJs on. However, there is a trade-off. Navigating through a professional event such as #VascularDisovery20 is challenging by itself, let alone going after it when you are not physically present. The following tips will help you to turn this year into a great opportunity.
1: Networking with Peers and Mentors: Online Presence
Now that there is no opportunity to join a table for lunch, mingle during the dinner event or grab a coffee with your colleagues from other countries, increase your online presence by interacting with attendees on Twitter. Use the chat mode during virtual sessions to ask questions or chat with others. If you have a burning question about a talk, either tweet at the presenter or email them. This is a great practice for increasing your “professional” online activity.
2: Utilize the Home Stay
Now that you have a desk in front of you instead of sitting on conference room chairs (which are really uncomfortable from time to time), take notes with ease, have your screen open with relevant papers to the talk, take high-quality images from the slides you find important and download the available contents in advance from the #HeartHub. In addition, you can now have a comprehensive look at your favorite posters and get connected with the presenters.
3: Plan Ahead
Although you may think that you can easily jump to the laptop and login to the talk that is “live”, the reality may be different. There are still concurrent sessions that you need to choose which one you attend. Also, there is always a possibility of a technical problem, so make sure that everything is set and you know exactly which talk you are going after. Also, pay attention to the time-zone listed in the schedule.
4: Hangouts at Conference Evenings: Say Hello to Face Time or Zoom
It is an unwritten tradition that many attendees get together after a long day of scientific endeavors to sit down, chit chat and grab a drink. Use the ATVB Journal virtual happy hour with EIC Dr. Alan Daugherty as an example. If you would like to hang out with the “conference buddies”, reach out to them, set up a private virtual meeting and catch up. This is a great practice to break those shyness barriers, especially if you are at the early career stage.
It is obvious that nothing compares to be physically present among your peers, colleagues and mentors. However, during these uncertain times, we can still manage to make the most out of the opportunities we are offered. As scientists, we are always learning to overcome new challenges and come up with new solutions, therefore, navigating through a virtual event not only is a fun challenge but also is a great learning experience with many opportunities.
“The views, opinions and positions expressed within this blog are those of the author(s) alone and do not represent those of the American Heart Association. The accuracy, completeness and validity of any statements made within this article are not guaranteed. We accept no liability for any errors, omissions or representations. The copyright of this content belongs to the author and any liability with regards to infringement of intellectual property rights remains with them. The Early Career Voice blog is not intended to provide medical advice or treatment. Only your healthcare provider can provide that. The American Heart Association recommends that you consult your healthcare provider regarding your personal health matters. If you think you are having a heart attack, stroke or another emergency, please call 911 immediately.”