In my previous blog post, I briefly discussed the importance of microbiome research and how to take the first steps in conducting a microbiome study. In today’s post, I will continue to discuss the importance of this area of research with a focus on obesity.
The worldwide epidemic of metabolic syndrome and obesity in different age groups both in the United States and around the world is considered as a major public health concern. Moreover, the human gut microbiome has been linked to metabolic disease and adiposity and it is not only a marker of disease, but also contributes to pathology.
Intestinal microbiota plays a critical role in the host metabolism and immune system that extents its related physiological functions to other organs including brains, liver and adipose tissue. Metagenomic-wide association studies indicate significant changes between gut microbiota metagenome of metabolically healthy versus unhealthy individuals. Such microbiota changes are thought to be a possible cause of obesity and therefore, intestinal microbiota represents a potential therapeutic target to manage obesity.
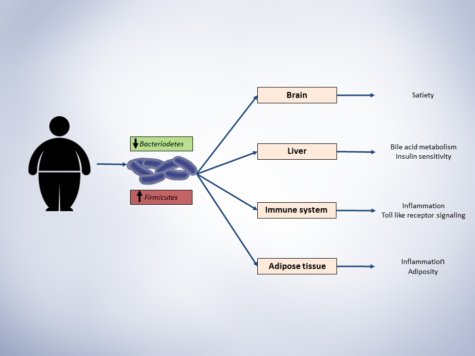
Overview of gut microbiota role in host metabolism. The shift in gut bacteria can affect host metabolism via several pathways in different tissues.
Study results have illustrated alterations in the dominant gut phyla of obese subjects/animals, reporting significant reduction in Bacteroidetes and significant increase in Firmicuts and Actinobacteria. The consequence of this shift in gut microbiota is the increased potential of harvesting energy from food and a low-level inflammation. Obesity leads to a low level inflammation, specifically in adipose tissue, that results in the production of several inflammatory cytokines, which may lead to insulin resistance as well. TNF-α, IL-1β and CCL2/MCP1 are among the important inflammatory cytokines that are induced in obese state accompanied by increased macrophages, T cells and mast cells. Presence of the aforementioned cells not only correlates with the gene expressions that control inflammation, but also indicates the possible role of innate immunity in obesity and insulin resistance. Moreover, Pattern recognition receptors such as Toll-like receptors (TRLs) are activated by bacterial endotoxins such as lipopolysaccharide (LPS), which results in innate immune response and inflammation. Also, gut microbiota produce wide range of molecules, such as flagellins and peptidoglycans, which activate inflammatory pathways leading to obesity and insulin resistance. Recent data from mice genetically deficient in TLR5 reported significant changes in their microbiota and development of metabolic syndrome characteristics. Results from germ free mice also illustrated possible effects of gut microbiota on host metabolism. High fat – high sugar diet fed mice did not show same metabolic disturbance in comparison with not germ free littermates. Microbiota transplantation from obese mice also resulted in greater adiposity in comparison with lean donor recipients. It is also suggested that short chain fatty acids (SCFAs) may contribute to regulation of gut dysbiosis. These compounds such as butyrate, acetate and propionate are produced by intestinal microbiota as a result of diet-derived fibers fermentation. SCFAs are thought to be the energy source for intestinal epithelium and liver, whereas they can also play a modulatory role in immune response via reducing gut permeability.

Shayan is a caffeine-dependent Ph.D. Candidate at the Saha Cardiovascular Research Center, University of Kentucky. His research area is focused on vascular biology and lipid metabolism. He tweets @MoradiShayan, blogs at shayanmoradi.com and he is the Winner of World’s Best Husband Award (Category: nagging).