Women in Electrophysiology
While I was chatting with a few fellows in our hospital hallway, I met one of the fellows who was very interested in electrophysiology (EP). We had a very interesting chat about her application and future career forward. In this blog, I summarize my chat with Jasneet Devgun, an aspiring electrophysiologist.
Question: Hi Jasneet, great to have you here! Let’s start with this question: When and how did you know you love EP?
- Answer: EP is something I never really thought of pursuing initially. In fact, I was interested in interventional cardiology since my second year of medical school. It was not until I met an electrophysiologist at the University of Chicago during my third year of medical school that I thought of EP as a possible future career. He was so excited to show me the world of EP and frequently took me to the lab to see EP in action. I still remember the day he said, “we need more women in EP…you should consider it.” From then on, my curiosity grew. I found myself drawn to the lab, scrubbing in on cases in residency and fellowship. The unique therapeutics, cutting-edge procedures and technology, intellectual and logical nature of EP, alongside very memorable and rewarding encounters with patients and wonderful attendings, made me realized that EP was the right field for me.
Question: This is great!! What are your thoughts about women in EP?
- Answer: Last year, Dr. Kamala Tamirisa wrote a very thoughtful piece for EP Lab Digest on the EP fellow shortage.1 At the time, the National Resident Matching Program (NRMP) demonstrated that approximately 40% of 130 EP fellowship positions in the US were unfilled.2 In 2021, that number drastically declined to 4%. Despite clear rising interest in EP, there remains a paucity of women in the field. The American Board of Internal Medicine (ABIM) reported that women comprised only 10% of first year EP fellows, while remaining steady at this rate for the past 10 years.3
The paucity of women pursing EP is a multi-faceted issue. A recent survey of cardiology fellows-in-training published in the Journal of American College of Cardiology showed that the most significant reasons women did not choose EP were greater interest in another field, radiation concerns, lack of female role models, a perceived “old boys’ club” culture, and discrimination/harassment concerns.4 Another reason was length of training. Reasons why women did choose EP were positive mentorship, unique features about the specialty, expertise, and the presence of a female role model, the latter being the major influencer.
These results are not surprising, but there are ways we can tackle the question at hand.
Question: Absolutely!! And this brings up the importance of mentorship, can you share your experience with that?
- Answer: I cannot stress enough the value of a good mentor. A good mentor inspires and cultivates the foundations of turning one’s future into reality. This was personally a huge factor for me; I did not know anything about, let alone consider, EP until I met electrophysiologists who had a genuine interest in my career development. Interestingly, none of them were female. Our male colleagues can be some of our biggest advocates. I certainly see how a female role model is uniquely relatable and valuable. However, the gap will remain until more females in EP exist. That said, networking with female electrophysiologists through existing organizations, as well as creating outreach/interest groups in-person and on social media to involve residents, medical students, and even undergraduate students, would be very effective.
Question: What advice do you have for fellows who do not know much about EP or are not sure if they would want to pursue it? What are some possible barriers to developing interest?
- Answer: Exposure is key! Many trainees do not have much exposure to EP, and therefore may not know enough to develop an interest for it. Fellows should be aware of the distinctive benefits and exciting features unique to EP, which can only be achieved by increasing their time with electrophysiologists and the EP lab. This can also dispel concerns about radiation safety. For example, female cardiology fellows concerned about radiation in the survey may not know about the use of 3D mapping systems and multi-modality imaging, affording “low-fluoro” or “fluoro-less” cases and reduction in procedure times. Moreover, the development of robotically assisted procedures has provided an avenue to reduce occupational hazards.
- I was fortunate that my residency program offered an EP elective, where I met electrophysiologists who were excited to show me their world. A structured elective early in residency and more electives for medical students, with some exposure to the lab, may help bridge the gap and channel early interest. The earlier it is, the better, since interests develop quickly (as did mine!).
Question: What are your thoughts on the length of training and how this may be impacting fellows’ wellness and career decisions?
- Answer: Length of training is an important issue.1,4 This time overlaps with childbearing years and critical family development. Fellows, both male and female, should not have to feel like they must choose one over the other. There must be a genuine culture of promoting work-life balance during the long years of training. Fellows are also consequently faced with prolonged financial strains from student loan debt. These are things that fellows consider when deciding to pursue another fellowship. As medicine progresses towards a milieu of sub-specialties requiring ever-more training, the training structure must be modernized to optimize the workforce. Unfortunately, many people, including female fellows, may be missing out on great sub-specialties like EP because of these issues. Some have proposed modifying the last 6 months of cardiology fellowship as the beginning of CCEP, which is a great short-term goal.1 A “fast-track” program in fellowship that may even extend into residency may be a proposition for much later in the future. These changes can make a major difference in fellows’ career decisions, health, and well-being.
Despite the paucity of women in EP, I am positive that the great strengths of this field will surpass any barriers to recruiting them. Building exposure early, having more visible role models and mentors, modification of the training structure, and many other solutions previously stated will allow for tremendous progress. Even simple interventions can make leaps and bounds in bringing more women into the wonderful world of electrophysiology.
I would like to thank Jasneet Devgun, DO, who is currently a general cardiology fellow at Henry Ford Hospital, and an aspiring electrophysiologist, for sharing her experience and thoughts with us. A special thank you goes to Dr. Judith Mackall and Dr. Cristina Tita who helped in writing this blog.
References:
- Tamarisa, K. The Importance of Choosing Cardiac Electrophysiology as a Career: Thoughts on the EP Fellow Shortage. EP Lab Digest. Available at https://www.hmpgloballearningnetwork.com/site/eplab/importance-choosing-cardiac-electrophysiology-career-thoughts-ep-fellow-shortage. Accessed October 9, 2021.
- Fellowship Match Data and Reports. National Resident Matching Program. Available at http://www.nrmp.org/fellowship-match-data/. Accessed October 9, 2021.
- Percentage of First-Year Fellows by Gender and Type of Medical School Attended. Available at https://www.abim.org/about/statistics-data/resident-fellow-workforce-data/first-year-fellows-by-gender-type-of-medical-school-attended.aspx. Accessed October 9, 2021.
- Abdulsalam N, Gillis AM, Rzeszut AK, Yong CM, Duvernoy CS, Langan MN, West K, Velagapudi P, Killic S, O’Leary EL. Gender Differences in the Pursuit of Cardiac Electrophysiology Training in North America. J Am Coll Cardiol. 2021 Aug 31;78(9):898-909. doi: 10.1016/j.jacc.2021.06.033. PMID: 34446162.
“The views, opinions, and positions expressed within this blog are those of the author(s) alone and do not represent those of the American Heart Association. The accuracy, completeness, and validity of any statements made within this article are not guaranteed. We accept no liability for any errors, omissions, or representations. The copyright of this content belongs to the author and any liability with regards to infringement of intellectual property rights remains with them. The Early Career Voice blog is not intended to provide medical advice or treatment. Only your healthcare provider can provide that. The American Heart Association recommends that you consult your healthcare provider regarding your health matters. If you think you are having a heart attack, stroke, or another emergency, please call 911 immediately.”
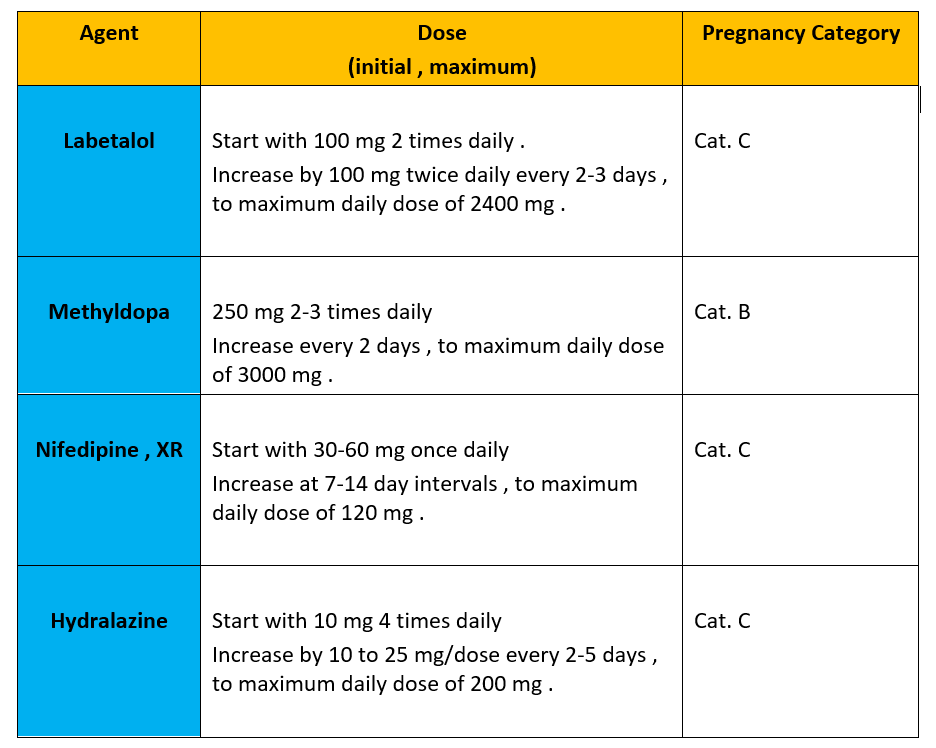
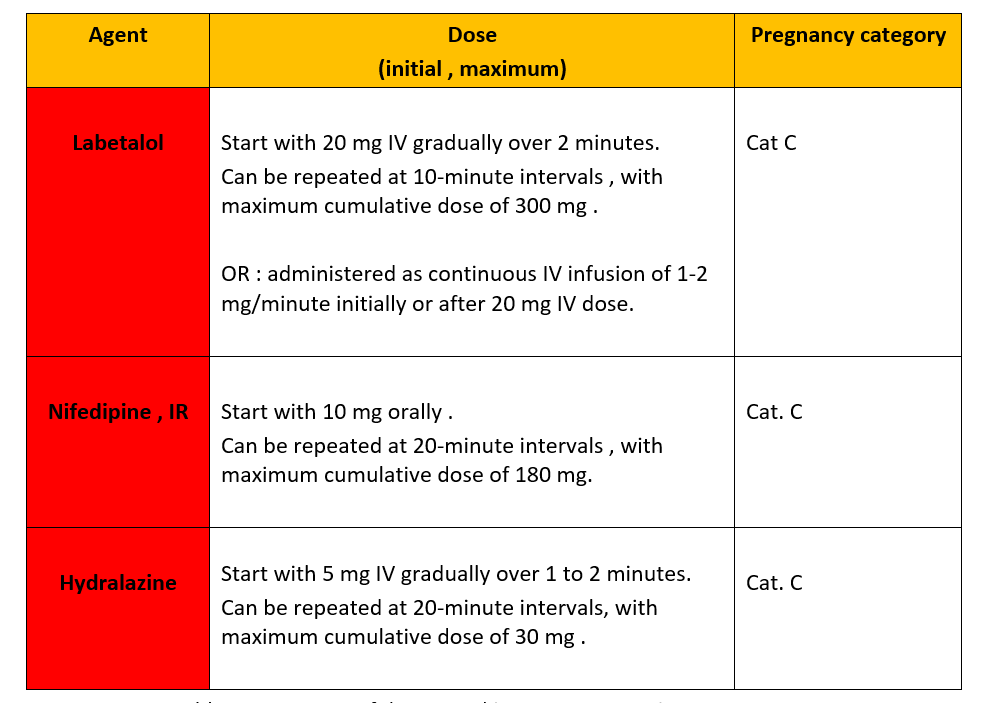
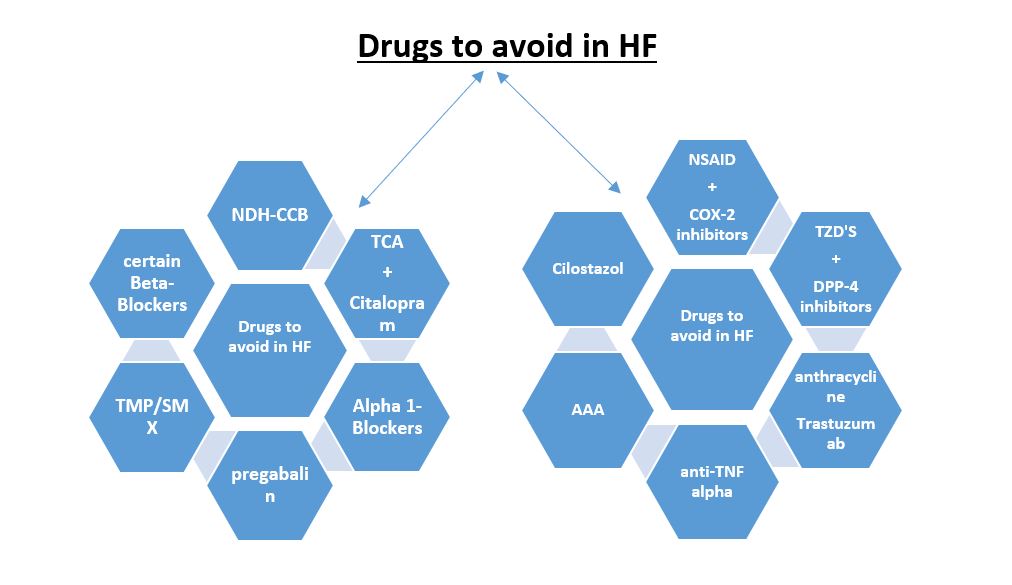
 It is February again of a new year of hope and progress!! Since it is the “The American Heart and Go Red for Women Month”, I would like to talk about the American Heart Association (AHA) GO RED initiative and discuss why heart disease in women is unique, urging my colleagues across the globe to work diligently to ensure optimal health and heart care for everyone, irrespective of their sex or gender.
It is February again of a new year of hope and progress!! Since it is the “The American Heart and Go Red for Women Month”, I would like to talk about the American Heart Association (AHA) GO RED initiative and discuss why heart disease in women is unique, urging my colleagues across the globe to work diligently to ensure optimal health and heart care for everyone, irrespective of their sex or gender.