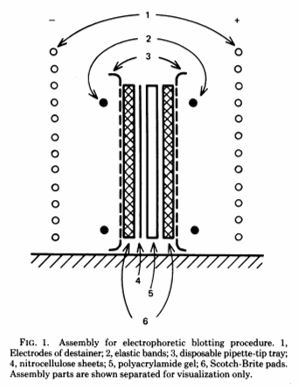
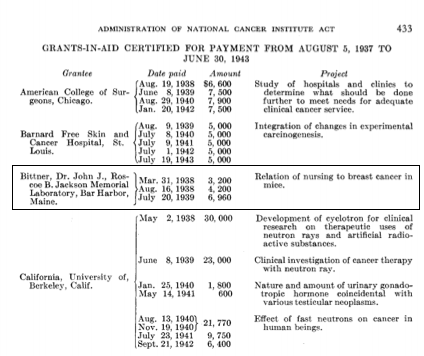
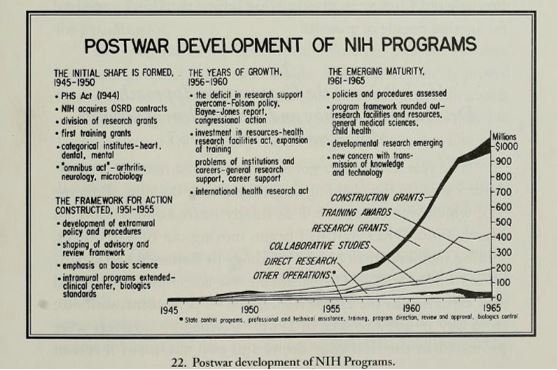
#BlackCardioInHistory: Daniel Hale Williams – Pioneer in open-heart surgery in the United States
This is part of the #BlackCardioInHistory series. #AHAEarlyCareerVoice is partnering with #BlackInCardio to feature a series of profiles of black/African American Cardiologists for #BlackInCardioWeek2020. For more information: blackincardio.com
Daniel Hale Williams III was born to a family that owned a barber business and worked in the Equal Rights League in Hollidaysburg, Pennsylvania, in 1856 (1). Young Daniel moved to Baltimore as a young boy to become a shoemaker’s apprentice when his father died but did not like the work (1). His family had moved to Illinois, so he moved back with them and began barbering with the long-term goal of pursuing his education (1).
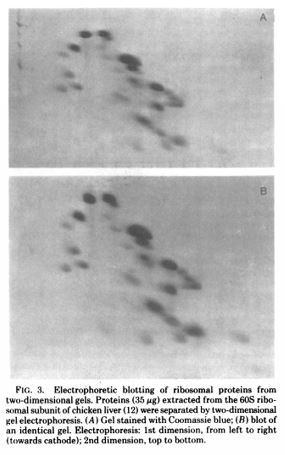
Daniel Hale Williams III apprenticed with Dr. Henry Palmer, who was an accomplished surgeon. From there, Daniel attended the Chicago Medical College (1).

(2) Daniel Hale Williams (Pre 1923 photograph, public domain). https://en.wikipedia.org/wiki/File:Daniel_Hale_Williams.jpg From Wikipedia page: Daniel Hale Williams.
After completing medical school, Dr. Williams began to practice on Chicago’s south side while teaching anatomy at the Chicago Medical College (1). He was an early adopter of Louis Pasteur’s sterilization to prevent transmission of infection (1).
In the late 1880s in America, Black Americans were prevented from being admitted to hospitals and could not be hired at hospitals (1). While Black women had a long history of working as nurses, they were often denied opportunities for formal training (3). Dr. Daniel Hale Williams III disagreed with this practice and opened the Provident Hospital and Training School for Nurses which was the first hospital in the United States that was racially integrated (1).

(3) Provident Hospital and Training School for Nurses (36th and Dearborn Streets, Chicago). Chicago History Museum (ICHi-040212). https://www.chicagohistory.org/provident-hospital/. Brittany Hutchinson.
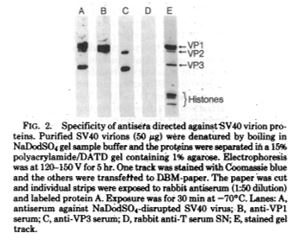
A few years later, in 1893, Dr. Williams treated a man with a significant stab wound to the chest, named James Cornish (1). Dr. Williams successfully sutured the man’s pericardium allowing Cornish to live for many years. Only 3 other surgeons, Dr. Francisco Romero, Dr. Henry Dalton, and Dr. Dominique Jean Larrey, have been credited as performing open-heart surgery previously (1,4). One year later, in 1894, Dr. Williams was appointed as Chief Surgeon of the Freedmen’s Hospital, which has seen a high relative mortality rate. He moved back to Chicago upon marrying Alice Johnson, where he continued working for Provident Hospital (1).

(2) Daniel Hale Williams (National Library of Medicine believes this to be public domain). https://commons.wikimedia.org/wiki/File:Daniel_H._Williams.jpg From Wikipedia page: Daniel Hale Williams.
In 1897 Dr. Williams was appointed to the Illinois Department of Public Health to increase medical standards in hospitals (2). In 1913, Dr. Williams was the only Black American member of the American College of Surgeons (5). Dr. Williams died in 1931 in Idlewild, Michigan from a stroke (2). Some of Dr. Williams’s honors include membership in the Chicago Surgical Society and American College of Surgeons, which were both uncommon for Black Americans at the time (2). Dr. Williams also received honorary degrees from Howard University in Washington DC and Wilberforce University in Wilberforce, Ohio (2).
Reference
- Biograhy.com Daniel Hale Williams Biography. 6/5/2020. https://www.biography.com/scientist/daniel-hale-williams. Accessed 10-19-2020.
- Daniel Hale Williams (Pre 1923 photograph, public domain). https://en.wikipedia.org/wiki/Daniel_Hale_Williams#cite_note-EB-6 From Wikipedia page: Daniel Hale Williams
- Provident Hospital and Training School for Nurses (36th and Dearborn Streets, Chicago). Chicago History Museum (ICHi-040212). https://www.chicagohistory.org/provident-hospital/. Brittany Hutchinson.
- Wikipedia.com Henry Dalton. 4/4/2020. https://en.wikipedia.org/wiki/Henry_Dalton. Accessed 10-19-20
- Daniel Hale Williams American Physician. Encyclopedia Britannica. 2018.
“The views, opinions and positions expressed within this blog are those of the author(s) alone and do not represent those of the American Heart Association. The accuracy, completeness and validity of any statements made within this article are not guaranteed. We accept no liability for any errors, omissions or representations. The copyright of this content belongs to the author and any liability with regards to infringement of intellectual property rights remains with them. The Early Career Voice blog is not intended to provide medical advice or treatment. Only your healthcare provider can provide that. The American Heart Association recommends that you consult your healthcare provider regarding your personal health matters. If you think you are having a heart attack, stroke or another emergency, please call 911 immediately.”