Everybody has a western blot horror story. The history of the western blot. What has been your worst western blot experience?
Most basic scientists have done countless western blots in the lab. The western blot, or SDS PAGE and immunoblot, technique is indispensable in most labs as a method to measure relative expression level of your protein of interest. As an early career scientist, I’ve done hundreds of western blots, and I often wonder who came up with this technique.
When I was learning this technique in grad school, I was told that it is called a “western” blot because the precursor techniques for detecting DNA and RNA in gels were called the “southern” blot and “northern” blot. The southern blot being named after Edwin Southern.
This was actually very easy to find out. I went to the references section of the “Western Blot” Wikipedia page and found the first citation for the technique (not yet called a western blot). There were two papers published in 1979 on the technique by different groups. The first was by Jaime Renart, Jakob Reiser, and George Stark in July. The second paper by Harry Towbin, Theophil Staehelin, and Julian Gordon was published in September, and both papers were published in the Proceedings of the National Academy of Sciences of the USA.1,2
According to PNAS, the Towbin paper has over 43,000 citations while the Renart paper has only 493. Both papers undoubtedly have had a much wider impact but most current papers do not cite these originals anymore. A pubmed search for the terms “western blot” or “protein immunoblot” returns a combined 130,000 papers.1,2
The main difference between these two manuscripts was that the Renart technique transferred the proteins from the SDS gel into paper whereas the proteins were transferred into nitrocellulose in the Towbin paper. Now nitrocellulose is used and many use PVDF membranes; however, different labs have different preferences.
The original assembly from the Towbin paper was very simple and not much different from the more sophisticated apparatus marketed by biotech companies today.
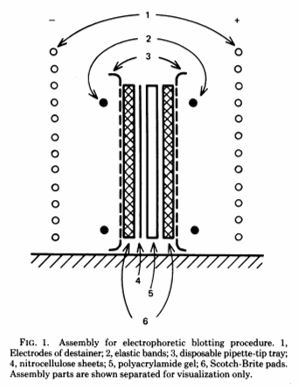
Towbin H, Staehelin T, Gordon J. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350-4.
The Renart paper shows blots probed with specific antisera and they look remarkably like blots shown in manuscripts today. The Towbin paper is remarkable because the authors then go on to show a variety of ways this technique can be used. They ran a 1-dimensional gel and stained all of the protein, in this case 60-S ribosomal proteins from chicken liver. Then they ran a 2-dimensional gel (shown below) of the same 60-S ribosomal proteins from chicken liver. Finally, they used antibodies to detect specific proteins and they demonstrated this using both horseradish peroxidase (HRP) and fluorescein (FITC).
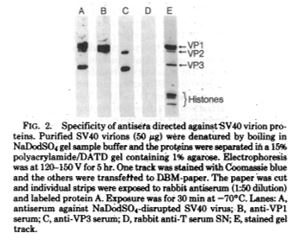
Renart J, Reiser J, Stark GR. Proc Natl Acad Sci U S A. 1979 Jul;76(7):3116-20.
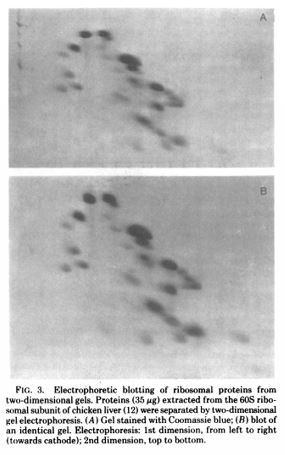
Towbin H, Staehelin T, Gordon J. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350-4.
This technique was officially coined “western blotting” in 1981 in a paper by W. Neal Burnette.3 While the name of the western blot is not very important, the technique is obviously one of the most valuable tools for molecular biologists, and one that provides constant amusement (or frustration) for graduate students and post-docs worldwide.
I have many of my own tips and tricks for western blotting but I won’t list them here. Everyone has their own western blot horror stories. One of my worst experiences is mixing up the red and black wires and seeing my precious samples running up and out of the wells, and if you use pre-cast gels which are available from several suppliers, please be kind to them. Don’t squeeze them:

Daniel Tyrrell, 2018, LC3 I and II western blot
References:
- Renart J, Reiser J, Stark GR. Proc Natl Acad Sci U S A. 1979 Jul;76(7):3116-20.
- Towbin H, Staehelin T, Gordon J. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350-4.
- Burnette WN. Anal Biochem. 1981 Apr;112(2):195-203.