The ISCHEMIA trial definitely caused quite a chatter, and congratulations to the authors on a thought provoking and interesting study. I’m fascinated and can’t wait to do a deep dive on my own time. BUT, that’s for stable ischemic heart disease. There’s a time and a place for that, but that place is certainly not the ICU.
With that in mind, I’m going to briefly highlight a few presentations at this year’s AHA conference, and throw out a few breadcrumbs to pique your interest and hopefully encourage you to look into the primary literature.
Utilization and Outcomes of Impella vs IABP Among Patients with AMI Complicated by Cardiogenic Shock Undergoing PCI
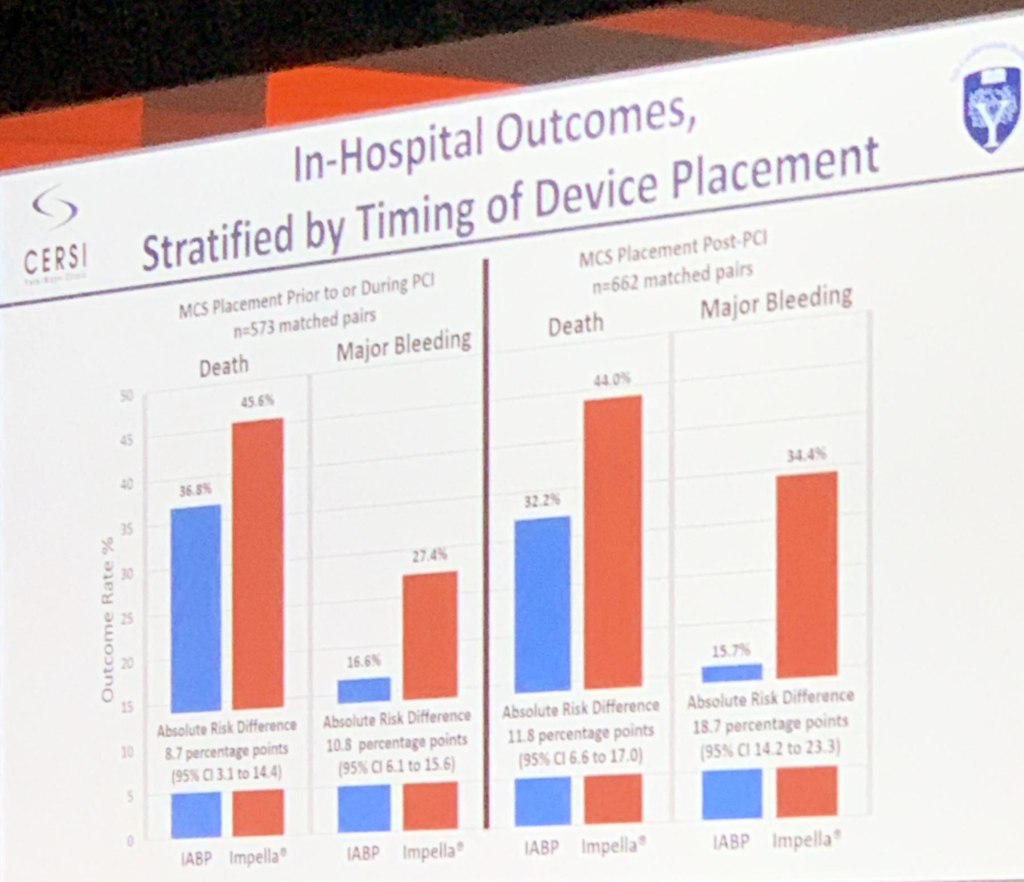
In this talk, Dr. Sanket Dhruva and friends took data from the NCR CathPCI Registry, and created a linked cohort with the Chest Pain MI Registry. Out of 28,304 patients with AMI and CS, 8,471 received IABP only, 1,768 received Impella only. They were able to propensity match 1,680 (95%) of the Impella patients to IABP patients. The results were staggering: 45.0% mortality rate for Impella vs 34.1% with IABP. Major bleeds (as defined by the NCDR) occurred in 31.3% of Impella and 16.0% of IABP patients. They also stratified the patient event rates for pre and post-PCI initiation of mechanical circulatory support. Study perior was 10/2015 – 12/2017
Pre-PCI initiation of MCS:
- Mortality 45.6% vs 36.8% Impella vs IABP
- Major bleeding 27.4% vs 16.6% Impella vs IABP
Post-PCI initiation of MCS:
- Mortality 44.0% vs 32.2% Impella vs IABP
- Major bleeding 34.4% vs 15.7% Impella vs IABP
Comments:
This was a historical cohort, this is observational data, and I think this highlights a need for a prospective RCT looking at the use of Impella vs IABP in patients with AMI and cardiogenic shock. Because of the nature of the dataset, the definitions of events were standardized across the board. I will also add that these patients were not stratified using the SCAI schema for cardiogenic shock (hard to use it when it wasn’t released during the study period!). Is it possible that the sicker patients got Impella more frequently? Sure, but, Dr. Dhruva noted that most of the Impella devices used were the small 2.5, and not the 5.0. A study I was a part of looking at trends in utilization and mortality with MCS published earlier this year also found similarly increasing trends in utilization of MCS, but no difference overall with regards to mortality. That paper, too, performed retrospective data analysis, and carries many of the same limitations as this study.
1 year outcomes of the COACT: Coronary Angiography after Cardiac Arrest Trial
You are the physician on-call at your ICU. You hear of a patient who had an out-of hospital cardiac arrest, the rhythm was VT, ROSC was obtained in the field, and the post-ROSC ECG showed no STEMI. Do you take them to the cath lab, or do you just focus on targeted temperature management & stabilize the patient? What if it was an electrically silent MI!?
This study sought to provide some guidance for scenarios such as the one above. Followup was assessed at 90 and 365 days for the 538 patients enrolled in this study.
Exclusion criteria was STEMI, obvious non-coronary cause of arrest, delivery of electric shock
Inclusion criteria: Initial shockable rhythm, comatose after ROSC (GCS < 8) , and no ST elevation on post-ROSC ECG.
End-result, 61.4% of patients in the immediate angiography and 64.0% in the delayed angiography group were alive 1 year post-arrest. No significant difference in rates of revascularization, MI, or hospitalization for heart failure or ICD shocks between either group.
Comments:
Looking at the details, I saw roughly similar rates of PCI in both groups, I am curious if PCI utilizing FFR would have had different results in the delayed angiography group. Overall, I think this provides solid evidence to support the importance of focusing on stabilizing the post-cardiac arrest patient, and looking for other, non-cardiac causes of the arrest (assuming no obvious signs of MI).
Session on Hemodynamics in Cardiogenic Shock
This has a lot of buzz words that I like, and I’m a huge geek for physiology, so I basically camped outside the room in anticipation of this session.
The speakers were an all-star lineup, including Dr. Hall from Baylor University, Dr. van Diepen from University of Alberta, Dr. Wong of the University of British Columbia, Dr. Kapur from Tufts, and Dr. Thiele from the University of Leipzig.
My breadcrumb summary:
- The old definition of cardiogenic shock is not very helpful (sustained SBP < 90 mm Hg for at least 30 minutes and CI < 1.8 L/min/m2 along with elevated fillings pressures of LV, RV, or both).
- The newly released scheme from the SCAI is more useful, and helps to better standardize and stratify patients across the spectrum of CS (and this was already validated in a huge cohort).
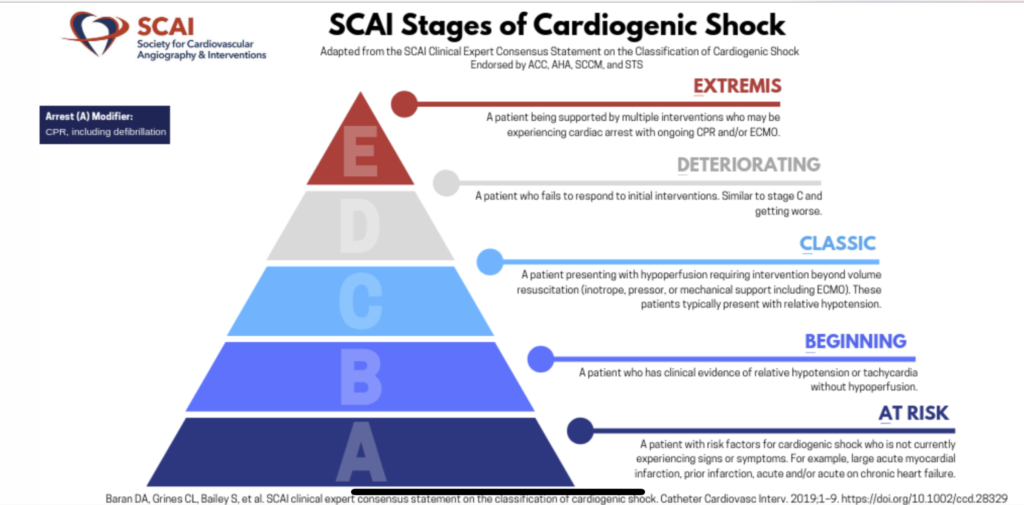
- The duration and type of CS matters! Cardiogenic shock in a patient with ADHF is different than the patient with acute myocardial infarction!
- When considering what device to use for MCS, it is important to consider a few things, namely, what is your center most familiar (and therefore best) at doing? How much volume/flow do you anticipate needing?
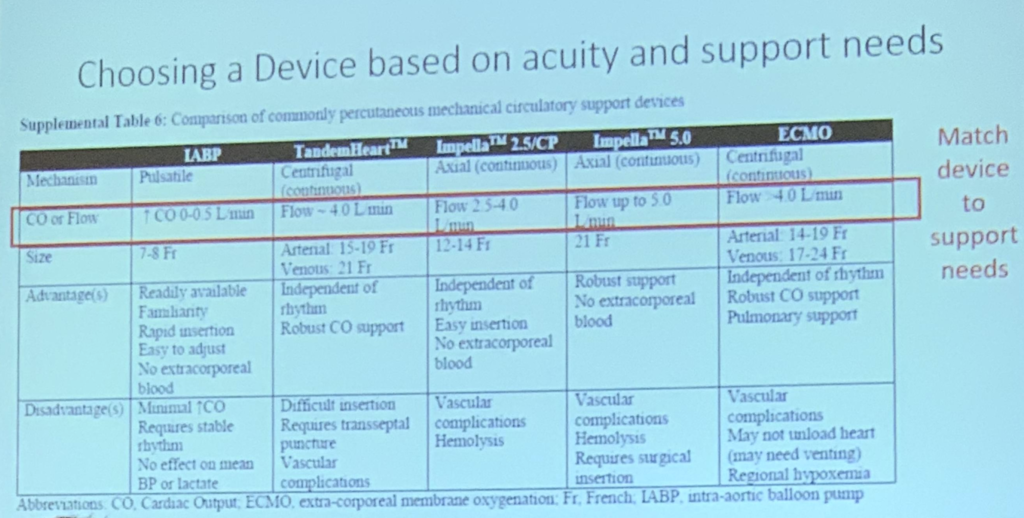
- The existing literature regarding MCS and CS associated with AMI is lacking in sample size, and we need more data.
Neurologic Function and Outcome After Cardiac Arrest
I’m not an expert, so I’m going to shamelessly regurgitate what the experts said. Namely, multi-modal imaging approaches can, with good specificity (but poor sensitivity), prognosticate poor chances of neurologic recovery 0 a very meaningful endpoint for these patients.
Similarly, there are a few biomarkers that may serve as standard serum measurements in the future, as we study them more.
Lastly, intranasal evaporative cooling (sounds super cool doesn’t it?) is a method of targeted temperature management that can improve neurologic outcomes after cardiac arrest.

Check my references below for more reading, I highly recommend doing a deep dive, as I could not possibly do these excellent physicians justice on their expert summaries.
References:
- Dhruva SS. Utilization and outcomes of Impella vs IABP among patients with AMI complicated by cardiogenic shock undergoing PCI. Presented at: AHA 2019. November 17, 2019. Philadelphia, PA.
- Panhwar, Muhammad Siyab, et al. “Trends in the Use of Short-Term Mechanical Circulatory Support in the United States–An Analysis of the 2012–2015 National Inpatient Sample.” Structural Heart (2019): 1-8.
- Lemkes J.One Year Outcomes of Coronary Angiography After Cardiac Arrest. Presented at: AHA 2019. November 17, 2019. Philadelphia, PA.
- Lemkes JS, Janssens GN, van der Hoeven NW, et al. Coronary Angiography After Cardiac Arrest Without ST-Segment Elevation. N Engl J Med 2019;380:1397-407.
- Schrage, Benedikt, et al. “Impella Support for Acute Myocardial Infarction Complicated by Cardiogenic Shock: Matched-Pair IABP-SHOCK II Trial 30-Day Mortality Analysis.” Circulation10 (2019): 1249-1258.
- Thiele, Holger, et al. “Intraaortic balloon support for myocardial infarction with cardiogenic shock.” New England Journal of Medicine14 (2012): 1287-1296.
- Van Diepen, Sean, et al. “Contemporary management of cardiogenic shock: a scientific statement from the American Heart Association.” Circulation16 (2017): e232-e268.
- Baran, David A., et al. “SCAI clinical expert consensus statement on the classification of cardiogenic shock: This document was endorsed by the American College of Cardiology (ACC), the American Heart Association (AHA), the Society of Critical Care Medicine (SCCM), and the Society of Thoracic Surgeons (STS) in April 2019.” Catheterization and Cardiovascular Interventions(2019).
- Jentzer, Jacob C., et al. “Cardiogenic shock classification to predict mortality in the cardiac intensive care unit.” Journal of the American College of Cardiology17 (2019): 2117-2128.
- Nordberg P, Taccone FS, Truhlar A, et al. Effect of Trans-Nasal Evaporative Intra-arrest Cooling on Functional Neurologic Outcome in Out-of-Hospital Cardiac Arrest: The PRINCESS Randomized Clinical Trial. 2019;321(17):1677–1685. doi:https://doi.org/10.1001/jama.2019.4149
The views, opinions and positions expressed within this blog are those of the author(s) alone and do not represent those of the American Heart Association. The accuracy, completeness and validity of any statements made within this article are not guaranteed. We accept no liability for any errors, omissions or representations. The copyright of this content belongs to the author and any liability with regards to infringement of intellectual property rights remains with them. The Early Career Voice blog is not intended to provide medical advice or treatment. Only your healthcare provider can provide that. The American Heart Association recommends that you consult your healthcare provider regarding your personal health matters. If you think you are having a heart attack, stroke or another emergency, please call 911 immediately.