Innovations in 3-D and 4-D Technology in the Cath Lab
There have been tremendous advances in 3-dimensional (3-D) technologies in the past few years, not only in various medical and surgical fields but also in our daily lives outside of work; with more and more new features in cell phones, computer design programs, and movies!! 4-dimensional (4-D) imaging captures 3-D images over time. These technologies are particularly important in cardiology, especially in interventional cardiology. The heart is a very dynamic organ, and understanding the variation in the anatomy of vessels and geometry of cardiac structures is key to ensuring successful procedures, patient’s safety and good outcomes. More recently, newer innovations in both 3-D and 4-D technologies have been developed, so I decided to shed light on some of these innovations and how they can be potential game-changers in the cath lab.
- 3-D Holograms
This technology was actually displayed at the Transcatheter Cardiovascular Therapeutics (TCT) 2019 meeting. It converts live transesophageal echo (TEE) imaging into real-time 3-D holographic video in the cath lab to aid structural heart procedures. The 3-D hologram is projected on a special display screen, and the interventional cardiologist uses hand movements and a foot pedal/switch to change the image orientation without breaking the sterile field. It also allows the operator to see the tools they use in the cath lab, including catheters or devices, in real-time in a 3-D format. This technology does not even require the user to wear 3-D glasses! It was submitted for FDA regulatory review in September 2019.
- HeartFlow Planner
This is a noninvasive, real-time virtual tool for coronary artery disease intervention. It allows interventional cardiologists to virtually map vessels on a 3-D coronary tree, with color codes indicating the fractional flow reserve-computed tomography (FFR-CT) values for each vessel as measured by a computational fluid dynamics algorithm. This seems to be a good tool for percutaneous coronary intervention (PCI) planning in vessels with significant disease; as it aims to provide us with a non-invasive way to determine whether a stenotic lesion if potentially flow limiting. However, it is important to note the CT-FFR has its own limitations, and some patients might still need invasive FFR for accurate assessment. This tool was approved by the FDA in September 2019.
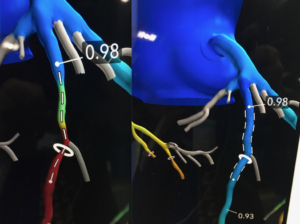 Figure 1: 3-D CT-FFR coronary tree showing both flow limiting and non-flow limiting lesions [from reference 1].
Figure 1: 3-D CT-FFR coronary tree showing both flow limiting and non-flow limiting lesions [from reference 1].
- 3-D Printing
3-D printing has been used in the surgical fields for more than a decade. It refers to making complex 3-D objects from computer-aided designs. This technology has been increasingly utilized in structural heart procedures in the past few years, where these 3-D models can be printed from a patient’s CT, magnetic resonance imaging (MRI), or 3-D ultrasound images (Figure 1). These 3-D printed structures not only help with procedural planning and device sizing but also allow operators to practice dry runs and perform pre-procedural navigation.
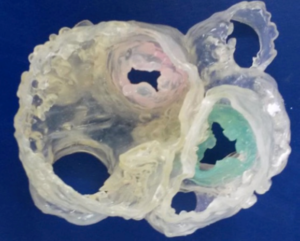
Figure 2: Image of a 3-D printed model which shows cardiac valves and major vessels with their geometric locations relative to each other (reference 3).
- 4-D Imaging
4-D imaging adds an important component to 3-D imaging, which is the change of these 3-D images over time. 4-D flow images include the direction of blood flow, blood velocities and shear wall stress [2] (Figure 3). This is particularly important in coronary interventions, structural heart procedures and different congenital abnormalities where identification of blood flow in the 4-D view is useful, especially when the anatomy is complex. These changes in position over time help guide our procedures, not only to ensure successful outcomes but also to avoid potential complications. These 4-D images require large amounts of data, but they can be obtained from either cardiac MRI or computational fluid dynamics, which is a specialized area of mathematics and fluid mechanics in engineering [2]. 4-D imaging is still in its early phases, but it is another exciting advancement in our field.
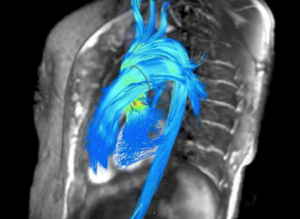
Figure 3: Representation of an MRI-generated 4-D flow image showing blood flow through the aorta and major vessels (reference 4).
In conclusion, we have seen and continue to see tremendous advances in the innovations of 3-D and 4-D imaging with important implications in our work in the cath lab. With our continued collaboration with informational technology experts, engineers, and scientists, these innovations are potentially game-changers in different fields, including coronary interventions and structural heart procedures. I look forward to seeing how this technology continues to evolve in the coming decades!!
References:
- Fornell, Dave “Overview of the top news and new technologies at the 2019 Transcatheter Cardiovascular Therapeutics meeting”, November 2019,
https://www.dicardiology.com/article/6-hot-topics-interventional-cardiology-tct-2019
- Warmerdam E, Krings GJ, Leiner T, et al. Three-dimensional and four-dimensional flow assessment in congenital heart disease. Heart 2020;106:421-426.
- Medgadget editors, “3D Printed Heart Model Made from CT Scans and 3D TEEs”, June 2015, https://www.medgadget.com/2015/06/3d-printed-heart-model-made-from-ct-scans-and-3d-transesophageal-echocardiography.html
- 4- Itn editors, “Arterys Showcases FDA-cleared 4D Flow MRI Software at RSNA 2016”, November 2016,
https://www.itnonline.com/content/arterys-showcases-fda-cleared-4d-flow-mri-software-rsna-2016
“The views, opinions and positions expressed within this blog are those of the author(s) alone and do not represent those of the American Heart Association. The accuracy, completeness and validity of any statements made within this article are not guaranteed. We accept no liability for any errors, omissions or representations. The copyright of this content belongs to the author and any liability with regards to infringement of intellectual property rights remains with them. The Early Career Voice blog is not intended to provide medical advice or treatment. Only your healthcare provider can provide that. The American Heart Association recommends that you consult your healthcare provider regarding your personal health matters. If you think you are having a heart attack, stroke or another emergency, please call 911 immediately.”